So, how big is a single water molecule? The quick answer is:
The diameter of a water molecule (H2O) is closely calculated to be about 0.000282 µm (micrometers – millionths of a meter) in diameter.
You may say this number (0.000282 µm) as: “Two-hundred-Eighty-Two millionths of a micrometer”.
Or you may move the decimal three places (0.282 nm) and say: “Two-Hundred, Eighty-Two-thousandths of a nanometer”.
Or you may move the decimal place another three places, so the number is whole (282.0 pm), and say:
“Two-Hundred, Eighty-Two picometers” or (282 trillionths of a meter).
Let’s Recap
0.000282 µm (micrometers – millionths of a meter) equals,
0.282 nm (nanometers – billionths of a meter) equals,
282.0 pm (picometers – trillionths of a meter).
Now that’s small!
The Oddly-Shaped Water Molecule


When describing the size of a water molecule, be aware that the shape of an individual water molecule is not a perfect sphere. The oxygen atom is flanked on either side by two hydrogen atoms at an angle of about 104.45 degrees.
The distance from the center of the oxygen atom to the center of one of the hydrogen atoms is about 95.84 pm (picometers – trillionths of a meter), which equals 0.0000958 µm (micrometers – millionths of a meter).
In 1967, the term “micron” was officially deprecated and officially no longer used synonymously with the term micrometer. The International System of Units (SI) revoked the term because it would’ve created confusion if paired up with the term “micro”, meaning millionths of a unit. Today, the term “micrometer” and the symbol μ is accepted internationally as the official term for millionths of a meter. Source: Wikipedia: Micrometre
Understanding Metric Prefixes for Measuring Small Things
These important prefixes are all in relation to the meter (metre is the British spelling):
milli – millimeter (mm) is one-thousandths of a meter – 0.001 meters (10-3)
micro – micrometer (µm) is one-millionth of a meter – 0.000 001 meters (10-6)
nano – nanometer (nm) is one-billionth of a meter – 0.000 000 001 meters (10-9)
pico – picometer (pm) is one-trillionth of a meter – 0.000 000 000 001 meters (10-12)
Converting Metric to the older Imperial system
The older Imperial system, where things are measured in feet, inches and pounds is unfortunately, still widely used in the United States. Unless you are a scientist, engineer or doctor, you’re likely most familiar with the old imperial system. Let’s understand the size of a water molecule in inches.
1 meter equals 39.36996 inches (1.09361 yard).
The size of a single water molecule in metric is 282 pm (two-hundred, eighty-two picometers).
282 x 10-12 => (0.000 000 000 282) times 39.36996 inches = 11.102-9 inches (0.000000011102 inches).
You may say this number as “eleven-point-one-hundred-two, billionths of an inch”. This is the size of a single water molecule (H2O) in inches.
Did I say small before!?
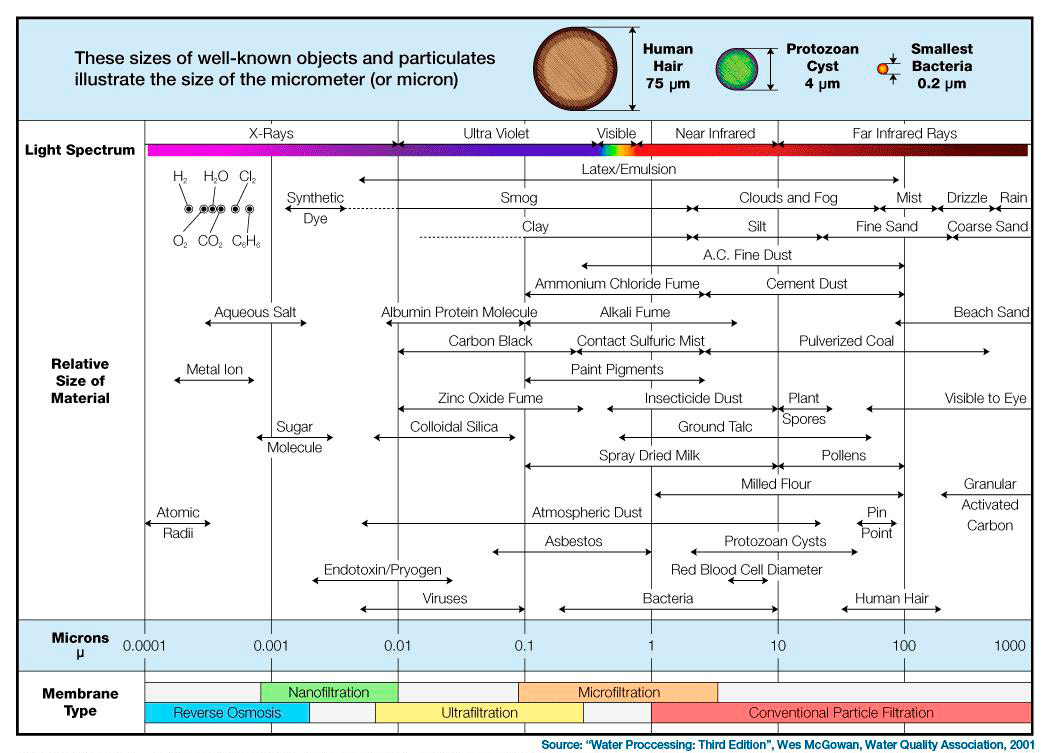
Note: See chart below where the water molecule (H2O) is located. The molecules CL2 is chlorine, and C6H6 is benzene, both toxic compounds. As you can see, these molecules are slightly larger than a water molecule.
More and more people are installing Home Reverse Osmosis Water Filter systems under their kitchen sinks, which purify their drinking water to eliminate these and other harmful chemicals.
The pores in an RO filter range in size from the smallest of 0.0001 µm to the largest pore size of about 0.0003 µm ( “three-ten-thousandths of a micrometer”). Most of these pores are just large enough for water molecules to squeeze through, but too small for larger contaminants to get through.
These impurities will not physically fit through the pores of a Reverse Osmosis filter. This physical, mechanical action is how contaminants are separated from your drinking water with an RO filter.
A Residential Reverse Osmosis system is your most effective way to remove the widest range of contaminants from your drinking water, making it safe to drink and cook with.
Click Chart to Enlarge
The human eye is capable of seeing particles down to about 40 microns without the use of a microscope. Use this micron particle size chart to give you perspective on particle sizes.